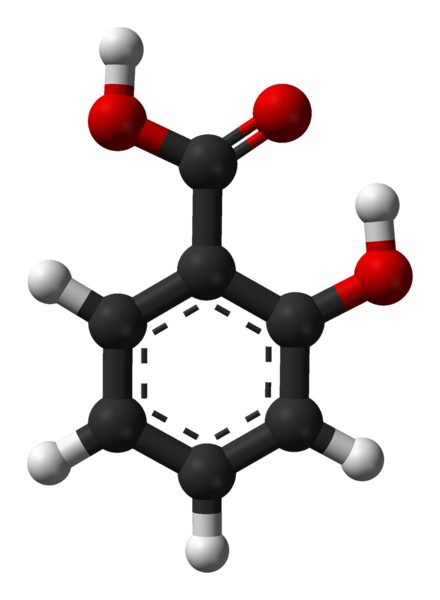
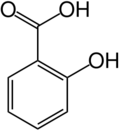
The chemical formula for salicyclic acid is C7H6O3 or HOC6H4COOH Salicylic acid is both a caroxylic acid and a phenol. A carboxylic acid is an organic (carbon-containing) acid characterized by the presence of a carboxyl group, which has the formula -C(=O)OH, usually written -COOH or -CO2H. A phenol, in the general sense of the term, is any compound which contains a six-membered aromatic ring, bonded directly to a hydroxyl group (-OH). In salicylic acid, the OH group is adjacent to the carboxyl group. Salicylic acid is only slightly soluble in water, but is soluble in ethanol and ether.

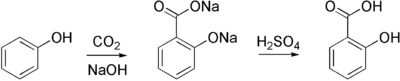
Salicylic acid is commercially prepared from sodium salicylate, which is produced from sodium phenoxide and carbon dioxide at high pressure and temperature in the Kolbe-Schmitt reaction. Sodium salicylate is acidified to give the desired salicylic acid:
A two-dimensional representation of the compound
A three-dimensional representation of the compound. The 3D structure is not experimentally determined, but computed by PubChem.